Fakultäten
Fakultäten
![]()
Combination of illumination and high resolution NMR spectroscopy: Key features and practical aspects, photochemical applications, and new concepts
P. Nitschke, L. Nanjundappa, R.M. Gschwind, Prog. Nucl. Magn. Reson. Spectrosc., 2019, 114-115, 86-134.
In the last decade, photochemical and photocatalytic applications have developed into one of the dominant research fields in chemistry. However, mechanistic investigations to sustain this enormous progress are still relatively sparse and in high demand by the photochemistry community. UV/Vis spectroscopy and EPR spectroscopy have been the main spectroscopic tools to study the mechanisms of photoreactions due to their higher time resolution and sensitivity. On the other hand, application of NMR in photosystems has been mainly restricted to photo-CIDNP, since the initial photoexcitation was thought to be the single key to understand photoinduced reactions. In 2015 the Gschwind group showcased the possibility that different reaction pathways could occur from the same photoexcited state depending on the reaction conditions by using in situ LED illumination NMR. This was the starting point to push the active participation of NMR in photosystems to its full potential, including reaction profiling, structure determination of intermediates, downstream mechanistic studies, dark pathways, intermediate sequencing with CEST etc. Following this, multiple studies using in situ illumination NMR have been reported focusing on mechanistic investigations in photocatalysis, photoswitches, and polymerizations. The recent increased popularity of this technique can be attributed to the simplicity of the experimental setup and the availability of low cost, high power LEDs. Here, we review the development of experimental design, applications and new concepts of illuminated NMR. In the first part, we describe the development of different designs of NMR illumination apparatus, illuminating from the bottom/side/top/inside, and discuss their pros and cons for specific applications. Furthermore, we address LASERs and LEDs as different light sources as well as special cases such as UVNMR(-illumination), FlowNMR, NMR on a Chip etc. To complete the discussion on experimental apparatus, the advantages and disadvantages of in situ LED illumination NMR versus ex situ illumination NMR are described. The second part of this review discusses different facets of applications of inside illumination experiments. It highlights newly revealed mechanistic and structural information and ideas in the fields of photocatalyis, photoswitches and photopolymerization. Finally, we present new concepts and methods based on the combination of NMR and illumination such as sensitivity enhancement, chemical pump probes, experimental access to transition state combinations and NMR actinometry. Overall this review presents NMR spectroscopy as a complementary tool to UV/Vis spectroscopy in mechanistic and structural investigations of photochemical processes. The review is presented in a way that is intended to assist the photochemistry and photocatalysis community in adopting and understanding this astonishingly powerful in situ LED illumination NMR method for their investigations on a daily basis.
__________________________________________________________________________________________________
LED-Illuminated NMR Spectroscopy: A Practical Tool for Mechanistic Studies of Photochemical Reactions
Y.Ji, D. A. DiRocco, J. Kind, C. M. Thiele, R. M. Gschwind, M. Reibarkh, ChemPhotoChem, Accepted Manuscript.
This Concept article highlights the development of a novel analytical tool, LED-NMR (a combination of in-situ light illumination using a light?emitting diode and NMR spectroscopy) and its variant UVNMR (LED-NMR coupled with UV/Vis absorption spectroscopy), as well as their applications in the mechanistic investigation of light?induced transformations. The utility of these new tools has been demonstrated by providing rich kinetic and structural data of reaction species offering mechanistic insights into photochemical and photocatalytic reactions. Furthermore, NMR actinometry has been recently developed as a practical and simple method for quantum yield measurements. Quantum yield is an important parameter in photo-induced processes, but is rarely measured in practice because of the barriers associated with traditional actinometry. These new tools and techniques streamline measurements of the quantum efficiency while affording informative mechanistic insights into photochemical reactions. We anticipate these techniques will enable chemists to further advance the rapidly emerging photochemistry field.

__________________________________________________________________________________________________
First CEST application in Photocatalysis to determine the sequence of intermediates!
Photoinitiated carbonyl-metathesis: deoxygenative reductive olefination of aromatic aldehydes via photoredox catalysis
S.Wang, L. Nanjundappa, J. Hioe, R. M. Gschwind, B. König, Chem. Sci., 2019, 10, 4580-4587.
Carbonyl–carbonyl olefination, known as McMurry reaction, represents a powerful strategy for the construction of olefins. However, catalytic variants that directly couple two carbonyl groups in a single reaction are less explored. Here, we report a photoredox-catalysis that uses B2pin2 as terminal reductant and oxygen trap allowing for deoxygenative olefination of aromatic aldehydes under mild conditions. This strategy provides access to a diverse range of symmetrical and unsymmetrical alkenes with moderate to high yield (up to 83%) and functional-group tolerance. To follow the reaction pathway, a series of experiments were conducted including radical inhibition, deuterium labelling, fluorescence quenching and cyclic voltammetry. Furthermore, NMR studies and DFT calculations were combined to detect and analyze three active intermediates: a cyclic three-membered anionic species, an α-oxyboryl carbanion and a 1,1-benzyldiboronate ester. Based on these results, we propose a mechanism for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond generation involving a sequential radical borylation, “bora-Brook” rearrangement, B2pin2-mediated deoxygenation and a boron-Wittig process.
C bond generation involving a sequential radical borylation, “bora-Brook” rearrangement, B2pin2-mediated deoxygenation and a boron-Wittig process.
__________________________________________________________________________________________________
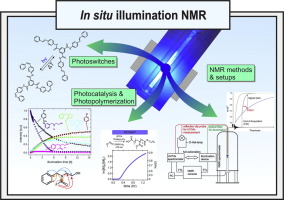
Combined In Situ Illumination-NMR-UV/Vis Spectroscopy: A New Mechanistic Tool in Photochemistry
A. Seegerer, P. Nitschke, R.M. Gschwind, Angew. Chem. Int. Ed., 2018, 57, 7493-7497.
Synthetic applications in photochemistry are booming. Despite great progress in the development of new reactions, mechanistic investigations are still challenging. Therefore, we present a fully automated in situ combination of NMR spectroscopy, UV/Vis spectroscopy, and illumination to allow simultaneous and time resolved detection of paramagnetic and diamagnetic species. This optical fiber based setup enables the first acquisition of combined UV/Vis and NMR spectra in photocatalysis, as demonstrated on a conPET process. Furthermore, the broad applicability of combined UVNMR spectroscopy for light induced processes is demonstrated on a structural and quantitative analysis of a photoswitch, including rate modulation and stabilization of transient species by temperature variation. Owing to the flexibility regarding the NMR hardware, temperature, and light sources, we expect wide ranging applications of this setup in various research fields.
__________________________________________________________________________________________________
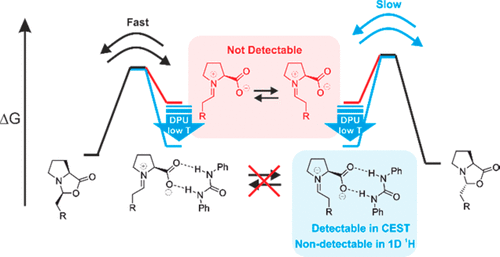
Chemical Exchange Saturation Transfer in Chemical Reactions: A Mechanistic Tool for NMR Detection and Characterization of Transient Intermediates
L. Nanjundappa, A. Seegerer, J. Hioe, R.M. Gschwind, J. Am. Chem. Soc., 2018, 140, 1855-1862.
The low sensitivity of NMR and transient key intermediates below detection limit are the central problems studying reaction mechanisms by NMR. Sensitivity can be enhanced by hyperpolarization techniques such as dynamic nuclear polarization or the incorporation/interaction of special hyperpolarized molecules. However, all of these techniques require special equipment, are restricted to selective reactions, or undesirably influence the reaction pathways. Here, we apply the chemical exchange saturation transfer (CEST) technique for the first time to NMR detect and characterize previously unobserved transient reaction intermediates in organocatalysis. The higher sensitivity of CEST and chemical equilibria present in the reaction pathway are exploited to access population and kinetics information on low populated intermediates. The potential of the method is demonstrated on the proline-catalyzed enamine formation for unprecedented in situ detection of a DPU stabilized zwitterionic iminium species, the elusive key intermediate between enamine and oxazolidinones. The quantitative analysis of CEST data at 250 K revealed the population ratio of [Z-iminium]/[exo-oxazolidinone] 0.02, relative free energy +8.1 kJ/mol (calculated +7.3 kJ/mol), and free energy barrier of +45.9 kJ/mol (ΔG?calc.(268 K) = +42.2 kJ/mol) for Z-iminium → exo-oxazolidinone. The findings underpin the iminium ion participation in enamine formation pathway corroborating our earlier theoretical prediction and help in better understanding. The reliability of CEST is validated using 1D EXSY-build-up techniques at low temperature (213 K). The CEST method thus serves as a new tool for mechanistic investigations in organocatalysis to access key information, such as chemical shifts, populations, and reaction kinetics of intermediates below the standard NMR detection limit.
__________________________________________________________________________________________________
1H DOSY Spectra of Ligands for Highly Enantioselective Reactions - A Fast and Simple NMR Method to Optimize Catalytic Reaction Conditions
K. Schober, E. Hartmann, H. Zhang, R. M. Gschwind, Angew. Chem. Int. Ed., 2010, 49, 2794-2797.
Phone: 0941 943-4626
Fax: 0941 943-4617
Room: CH23.1.80
E-Mail